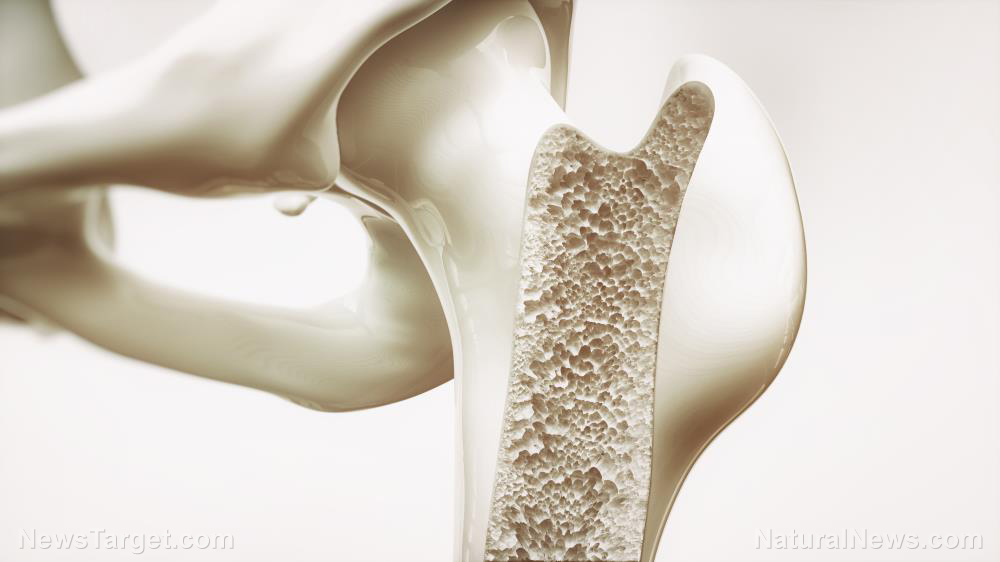
This mechanism appears to be more active in the ankles and less so in the hips. This, according to the researchers, is similar to the regenerative pattern seen in animals like salamanders. This finding could potentially lead to new treatments for osteoarthritis, the most common joint disorder worldwide.
Humans regenerate cartilage like salamanders
According to the researchers, humans are believed to be unable to counteract repetitive joint use and injury. This is because the turnover of insoluble collagen – a key component of the connective tissues – is thought to be limited in human adult cartilage.
For their study, the researchers examined protein turnover in the cartilage found in the lower limb joints of humans. To do this, they determined the age of proteins using internal molecular clocks that are integral to amino acids. Newly-formed proteins have fewer amino acid conversions compared to older proteins.
The researchers found that the cartilage tends to be young in the ankles, middle-aged in the knee and old in the hips. This pattern is also found in animals with regenerative abilities, where regeneration usually happens more actively at the tips of the body, including the ends of legs or tails.

Further analysis showed that cartilage regeneration in humans is likely regulated by molecules called microRNAs. These molecules were previously found to be more active in animals known for limb, fin or tail regeneration, such as salamanders, zebrafish, African freshwater fish and lizards.
Human microRNA activity is highest in the ankles compared to the knees and hips and higher in the top layer of the cartilage compared to the deeper layers. This finding may help explain why ankle injuries heal quicker and rarely become severely arthritic compared to knee and hip injuries. The researchers said that regulators like microRNAs could be potential targets for preventing and treating osteoarthritis. (Related: Compound in horny goat weed can promote cartilage repair.)
“We believe we could boost these regulators to fully regenerate degenerated cartilage of an arthritic joint,” said corresponding author Virginia Kraus of the Duke University School of Medicine.
The next step is to determine the regulators in salamanders that are not present in humans. These missing components, in turn, can provide the foundation for new approaches to repair joint tissues and possibly the whole human limbs, according to Kraus.
Regrowing cartilage using 2 proteins
In another study, published in the journal Nature Communications, researchers partially stimulated joint and bone growth in the amputated toes of mice using two proteins, BMP2 and BMP9.
The researchers previously regenerated bone in mice using BMP2. So for their study, they administered this protein alongside BMP9 to see their combined effects. Results showed that over 60 percent of the stump bones formed a layer of cartilage within three days.
The effect was strongest when BMP2 was applied first and BMP9 added a week after. Used this way, the proteins led to the growth of more complete joint structures, some of which even formed connections to the bone.
While further research is needed, the findings of these two studies could lead to new treatments for osteoarthritis that involve naturally replacing rather than transplanting cartilage.
Visit Prevention.news to read about treatments for osteoarthritis.
Sources include:
Please contact us for more information.