Medical meshes help a surgery patient recover faster after an operation that removed, repaired or replaced damaged tissue in his body. A mesh holds muscles tight and speeds up the healing process compared to sewing and stitching.
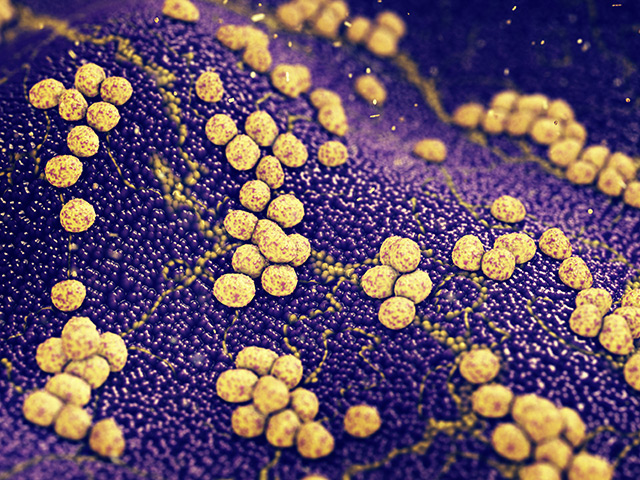
However, inserting a medical implant in a patient's body brings the risk of contamination by disease-causing bacteria. The microbes may cover the surface of the surgical mesh with an infectious biofilm.
A biofilm blocks any antibiotic agent from reaching the microorganisms below the coating. It reduces the effectiveness of antibiotic therapies against drug-resistant bacteria.
As a result of bacterial contamination, the patient may end up in repeated or even endless surgeries that may end in death. In 2015 alone, more than 30,000 people in Europe died from infections caused by antibiotic-resistant bacteria.
Healthcare professionals developed several methods to try and prevent bacteria from contaminating surgical implants. However, none of the approaches fully succeeded in stopping bacterial contamination of medical meshes. (Related: Treating wounds: How to recognize and prevent wound infection.)
A gold nanoparticle coating that generates heat in near-infrared light
Developed by researchers from the Institute of Photonic Sciences (ICFO) with help from the Catalan Institution for Research and Advanced Studies (ICREA), the new medical mesh featured a surface coated with gold nanoparticles. The tiny metal bits converted light into heat that filled their immediate surroundings.
Earlier studies have demonstrated the feasibility of light-heat conversion processes with gold nanoparticles. The approach promises a new way to treat cancer cells.
The researchers adapted the gold nanoparticle technique to medical meshes intended as surgical implants. Their approach might reduce the expenses of recurring surgical operations while also removing the need for costly yet ineffective antibiotic therapy.
In an in vitro experiment, the researchers applied millions of gold nanoparticles on the medical mesh. They made sure to spread the nanocoating across the entire surgical implant.
The researchers investigated the long-term stability of the gold nanoparticles, the material's ability to withstand degradation and the particles' ability to remain anchored onto the surface of the medical mesh.
Next, the researchers exposed the modified mesh to the pathogenic bacteria Staphylococcus aureus for 24 hours. They waited for a biofilm to appear on the surface.
A short but intense bombardment with pulses of near-infrared light brought the nanocoating of the medical mesh to thermal equilibrium. The researchers repeated the treatment 20 times with brief breaks between each pulse.
Killing antibiotic-resistant bacteria with heat while leaving healthy tissue alone
The ICFO-ICREA researchers reported that shining near-infrared light caused the gold nanoparticles to enter a mode called "localized surface plasmon resonance." The particles converted light into heat with high efficiency and effectively roasted the bacteria on the surface of the medical mesh.
Next, they evaluated the number of biofilm bacteria that died and survived the heating process. The surviving microbes transformed into planktonic cells that proved vulnerable to antibiotic therapy or the natural immune response.
Similarly, shining more light on the medical mesh made the dead bacteria less sticky. The lifeless microorganisms sloughed off the hot surface of the mesh.
Further, the researchers reported that medical mesh exposed to near-infrared light stayed compatible with in vivo settings. Thus, the bacteria-killing heat generated by the light-activated mesh would probably not harm nearby healthy tissue.
Lastly, the researchers repeated the near-infrared light treatment of the mesh. After analyzing the mesh, they verified that multiple heating did not degrade the gold nanoparticle coating's ability to convert light into heat.
Sources include:
Please contact us for more information.