Now, a new high risk population is being identified: Those who take anti-TB drugs are at an increased risk of re-infection. Even though current treatments such as isoniazid, rifampicin, and pyrazinamide are effective at controlling Tb in the short term, these drugs actually heighten the risk of re-infection. Drug resistant Tb is becoming a public health nightmare. The anti-TB drugs are making the problem worse in the long run. Now, researchers are figuring out why.
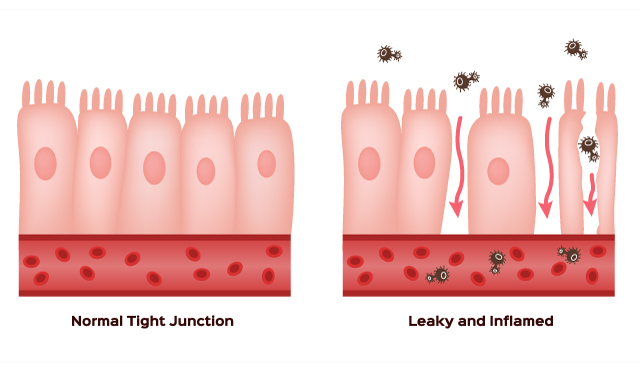
Anti-Tb drugs raise the risk of re-infection because these antibiotics fundamentally alter the microbes in the gut that protect against infections in the long run. A team of researchers from the Research Institute of the McGill University Health Centre published a study in Mucosal Immunology showing how anti-TB drugs alter the gut microbiota, leading to increased susceptibility to Mtb infection. The friendly microbes living in the human gut carry out important tasks including proper digestion, and immune system response. The anti-TB drugs damage this community of beneficial microbes, weakening the body’s ability to adapt to Tb and other infections in the long run.
Anti-Tb drugs disable protective microbes that communicate to macrophages in the lungs
Taking a closer look, lead researchers Dr. Irah King and Maziar Divangahi inundated mice for eight weeks with isoniazid, rifampicin and pyrazinamide, three commonly used anti-TB drugs. All three drugs altered the microbiome of the mice. The mice that were treated with a combination of isoniazid and pyrazinamide, showed increased susceptibility to Mtb infection. Going a step further, the researchers transplanted the feces from these mice into a new group of healthy mice that had not received anti-Tb drugs. The fecal transplant altered the microbiome of the new mice and compromised their immunity, making them more susceptible to Mtb infection.
Finally, the researchers investigated the gut-lung axis, where microorganisms communicate back and forth between the gastrointestinal tract and the lungs. A specific kind of immune cell located in the lungs, alveolar macrophages, are signaled to fight Mtb once the good bacteria detect the presence of this pathogen. If the good bacteria have been destroyed by anti-TB drugs, the macrophage response is not prompt or effective at all, allowing Mtb infection to take a stronger hold over the lung cells. It turns out that the friendly microbes are the intelligent first respondents to protect the body. When they are depleted, macrophages in the human immune system are not signaled to protect crucial organs. Anti-Tb drugs are disabling the microbiome’s ability to detect future infections and therefore allowing future infections to take a stronger hold over the patient’s lungs.
"We need to do more research in order to understand how the microbiome affects alveolar macrophages because these cells are critical for controlling early TB infection. We also need to identify the molecular pathways involved in the gut-lung axis,'' explains King. Future medical research should monitor changes in the human microbiome after antibiotics have destroyed it. Future medical research should investigate how to replenish patients’ microbiome quickly so that drug resistant Tb does not continue to destroy people’s lives.
Sources include:
Please contact us for more information.